A conductivity meter for water is an essential tool for measuring the ability of water to conduct electric current. This measurement reveals critical information about the ionic composition of the water. According to Dr. Jane Smith, a leading expert in environmental science, "A conductivity meter for water is crucial for assessing water quality and ecological health."
These devices provide insights into various water sources, including rivers, lakes, and tap water. Understanding conductivity is vital for monitoring pollutants and ensuring safe drinking water. Many environmental scientists rely on these meters to gauge the health of aquatic ecosystems.
However, not all conductivity meters are created equal. Factors like calibration, temperature, and maintenance can affect readings. It’s important to choose the right tool for specific applications. An unreliable meter can lead to misleading results, raising concerns about water quality assessments. As you explore the functionality of conductivity meters for water, consider how precision and accuracy impact environmental health.

A conductivity meter is a vital tool in water analysis. It measures the ability of water to conduct electricity. This ability is influenced by the presence of dissolved ions. Commonly, these ions include salts, minerals, and metals. Accurate conductivity measurements can indicate the overall quality of water.
In many industries, maintaining water quality is crucial. For example, agricultural studies indicate that optimal conductivity levels affect crop yield and health. Research shows that conductivity readings above 2,000 µS/cm can signal potential problems in drinking water supplies. The World Health Organization recommends monitoring these levels closely.
Many field professionals use conductivity meters regularly. Yet, there is often a lack of understanding regarding their calibration and maintenance. Without proper care, inaccurate readings may lead to misguided decisions. Often, meters go uncalibrated for extended periods. This oversight can have serious implications for water testing results. Awareness and training are essential for reliable data collection in water analysis.
Conductivity meters are essential tools in water testing. They measure the ability of water to conduct electrical current. This ability is influenced by the concentration of ions in the water, which can indicate the presence of various substances. Understanding these principles is vital for accurate water quality assessment.
When water contains dissolved salts or minerals, it becomes conductive. Higher ion concentration results in higher conductivity readings. For instance, freshwater typically has a conductivity range of 50 to 1500 µS/cm, while seawater can exceed 50,000 µS/cm. This data reflects the significant impact of ion presence on conductivity levels.
Tip: Always calibrate your meter before testing to ensure accurate readings. Also, consider temperature effects, as conductivity changes with temperature variations.
Measurement principles focus on the ratio of voltage to current. A conductivity meter applies a voltage between two electrodes submerged in the water sample. This allows for real-time monitoring of conductivity. However, the presence of organic materials can distort results. It’s important to be aware of these factors for reliable water analysis.
Tip: Regularly clean the electrodes to avoid contamination. This step enhances measurement accuracy during testing.
This bar chart illustrates the conductivity levels measured in various types of water, highlighting the significant differences in conductivity among distilled water, tap water, lake water, sea water, and wastewater. Conductivity values are expressed in microsiemens per centimeter (µS/cm), showing how different water sources can vary widely in their ionic content.
A conductivity meter is an essential tool for measuring the electrical conductivity of water. Its accuracy depends heavily on its key components. Understanding these parts can enhance your use of this instrument.
One crucial component is the sensor, which often consists of two electrodes. These electrodes measure the ionic content in water. When water is passed through, ions conduct electricity between the electrodes. The resulting conductivity value indicates the concentration of dissolved salts. Clean electrodes are vital for accurate readings. Ensure they are regularly maintained to prevent contamination.
Another important part is the display unit. It shows real-time conductivity readings and often includes temperature compensation features. This feature corrects the conductivity readings as temperature affects ionic mobility. Make sure to calibrate the device according to the manufacturer’s guidelines to sustain its accuracy.
**Tips**: Always store the conductivity meter in a safe, dry place after use. Regular checks on calibration can save time and resources. Keep a log of readings to track changes in water quality over time. Regular maintenance is just as crucial as the initial measurement.
| Component | Function |
|---|---|
| Probe | Measures the electrical conductivity of water |
| Microcontroller | Processes the conductivity data and converts it into readable units |
| Display | Shows the conductivity readings to the user |
| Calibration Standards | Ensures accurate measurements by calibrating the probe |
| Power Supply | Provides power to the meter for functionality |
| Temperature Sensor | Compensates for temperature variations affecting conductivity readings |
Conductivity meters are essential tools for assessing water quality. Understanding the accuracy of these devices hinges on calibration standards. Calibration ensures that measurements reflect true conductivity levels, which can influence agricultural practices, industrial processes, and environmental monitoring.
The precision of conductivity meters is often guided by standards set forth by organizations like ASTM International. For instance, ASTM D1125 outlines the procedures for calibration, indicating a need for regular testing against standard solutions. This helps minimize errors that may occur due to factors such as temperature fluctuations or sensor drift. Calibration solutions typically range from 0 to 100,000 µS/cm to accommodate various water types.
Despite significant advancements, challenges remain. Many users struggle with the proper calibration frequencies, sometimes leading to inaccurate readings. A 2021 study revealed that nearly 30% of tested meters were not calibrated correctly, impacting overall data reliability. This calls for more rigorous training and adherence to industry standards. Both experienced professionals and novices can benefit from reminders about calibration importance and operational best practices in maintaining accuracy in water conductivity testing.
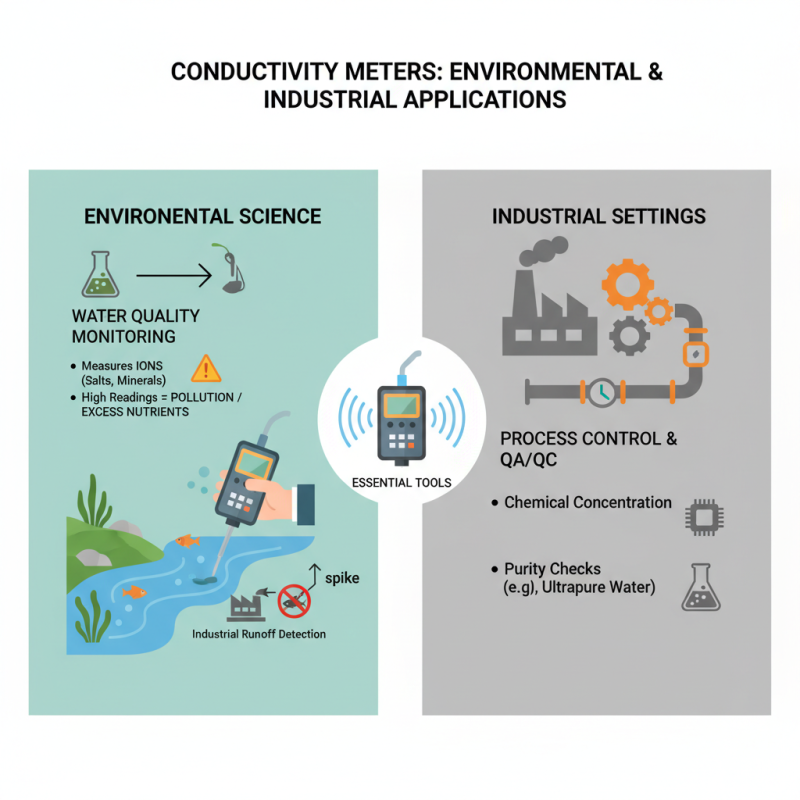
Conductivity meters are essential tools in various fields. In environmental science, they measure the electrical conductivity of water. This property helps determine the concentration of ions in water, such as salts and minerals. High conductivity readings can indicate pollution or excessive nutrient levels in aquatic systems. For instance, a sudden spike in conductivity might signal industrial runoff affecting local water bodies.
In industrial settings, conductivity meters play a crucial role in quality control. They ensure that water used in processes meets specific standards. In the semiconductor industry, purity is vital. Even minor impurities can lead to significant operational issues. Monitoring water conductivity helps maintain these standards. Regular calibration of the meters is necessary to ensure reliable data. However, not all readings are straightforward. Blind reliance on tools can sometimes lead to oversight of other crucial factors. Understanding the context of readings requires expertise.
Conductivity meters also have limitations. They may not distinguish between different types of ions. Thus, interpreting results can be complex. This calls for careful analysis beyond just numbers. A comprehensive approach can lead to better water quality management.